Half-Life and Radioactivity
Radioactivity and Half-Life
- Radioactive decay is when an unstable nucleus breaks apart into smaller pieces
- This occurs spontaneously, randomly, and naturally
- The nucleus gives off small particles
- Alpha particles (helium nuclei)
- Neutrons
- Protons (rare)
- Energy is also generally released
- Gamma (radiation only, no mass)
- Beta (electrons from just outside the nucleus)
- Some scientists involved in early research into radioactivity
- Henri Bacquerel, Ernest Rutherford, Paul Villard, Pierre Curie, Marie Curie
- Half-Life is the amount of time it takes for a material to decay by half
- EX: If you have 20 grams of a radioactive material and it decays to 10 grams in 4 days, then the half-life is 4 days. In another 4 days, there will be 5 grams left.
- Half-life is abbreviated as t½
- The unit for t½ depends on the material
- The amount of material remaining can be found in two ways
- Create and use a half-life chart
- Use the formula
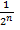 (n = number of half-lives)
(n = number of half-lives)
Positive Uses of Some Radioactive Elements
Isotope |
Use |
| 131I | diagnosis and treatment of thyroid disorders |
| 14C / 12C Ratio | dating once-living organisms |
| 236U / 206Pb Ratio | geological dating |
| 60Co | cancer treatment |
Half-Life Charts
| Number of Half-Lives | Amount of Substance Remaining | |
| As a Fraction | As a Percent | |
| 0 | 1/1 | 100% |
| 1 | 1/2 | 50% |
| 2 | 1/4 | 25% |
| 3 | 1/8 | 12.5% |
| 4 | 1/16 | 6.25% |
| 5 | 1/32 | 3.125% |
| 6 | 1/64 | 1.5625% |
Other Types of Problems
- You make one column for the number of half-lives
- Always start at zero.
- Make another column for amount of material
- This may be any form of “amount”
- Fraction (start at 1/1)
- Percent (start at 100%)
- Mass (start at the total mass)
- Amounts decrease BY HALF (divide by 2) as the number of half-lives increases
- This may be any form of “amount”
- Make another column for time passing
- Time can be in any unit
- Always start at zero.
- Time increases BY ITSELF
- Ex: if half-life is 3 days, then the times are 3, 6, 9, 12 …
- Ex: if half-life is 13 hours, then the times are 13, 26, 39 …
- Time can be in any unit
You have 68 grams of a radioactive material. It undergoes 4 half-lives. How much material remains?
| Number of Half-Lives | Amount of Substance Remaining | ||
| Fraction | Percent | Mass (g) | |
| 0 | 1/1 | 100% | 68 |
| 1 | 1/2 | 50% | 34 |
| 2 | 1/4 | 25% | 17 |
| 3 | 1/8 | 12.5% | 8.5 |
| 4 | 1/16 | 6.25% | 4.25 |
Determining the Number of Half-Life
- Determining the number of half-lives
- What you need:
- The half-life of the material
- The total amount of time
- What you need:
- Determining the amount of time passed
- What you need:
- The half-life of the material
- The number of half-lives
- What you need:
- Determining the amount of starting material
- What you need:
- The number of half-lives
- The amount of material you currently have
- What you need:
- Determining the half-life of the material
- What you need:
- The total amount of time
- The percentage (or fraction) of material you currently have
- What you need:
- There are other variations. You just need to reason them out.
- In most cases, a half-life chart will help
A radioactive material has a half-life of 23 days. If you measure it 207 days later, how many half-lives did it undergo?
- You can solve this more than one way:
- Create a half-life chart
- Use math
- total time divided by the half-life
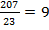 half-lives
half-lives
| # Half-Lives | Total Time (days) |
| 0 | 0 |
| 1 | 23 |
| 2 | 46 |
| 3 | 69 |
| 4 | 92 |
| 5 | 115 |
| 6 | 138 |
| 7 | 161 |
| 8 | 184 |
| 9 | 207 (stop here) |
Determining the Amount of Time Passed
A radioactive material has a half-life of 12.5 days. If it undergoes 6 half-lives, how much time passes?
- You can solve this more than one way:
- Create a half-life chart
- Use math
- half-life x # half-lives
- 12.5 x 6 = 75 days
| # Half-Lives | Total Time (days) |
| 0 | 0 |
| 1 | 12.5 |
| 2 | 25 |
| 3 | 37.5 |
| 4 | 50 |
| 5 | 62.5 |
| 6 | 75 |
Determining the Amount of Starting Substance
A radioactive material has a half-life of 8 days. If you have 13 grams after 32 days, how much was there in the beginning?
- You can solve this more than one way:
- Create a half-life chart
- Work down to find the # of half-lives, then work up to find the amount
- Use math
- Find out how many half-lives
- 32/8 = 4 half-lives
- Use 13 x 2n = 13 x 24 = 13 X 16 = 208 grams
- Create a half-life chart
| # Half-Lives | Total Time (days) | Amount of Material (grams) |
| 0 | ↓ 0 | 208 |
| 1 | ↓ 8 | ↑ 104 |
| 2 | ↓ 16 | ↑ 52 |
| 3 | ↓ 24 | ↑ 26 |
| 4 | 32 → | ↑ 13 |
Determining the Half-Life of a Material
You have 12.5% of a radioactive material remaining after 36 hours. What is the half-life of the material?
- You can solve this more than one way:
- Create a half-life chart
- Find the number of half-lives
- Divide total time by number of half-lives
- Use math
- 12.5% =
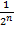 →
→
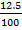 =
=
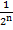 → 8 = 2n
→ 8 = 2n
- 8 = 2n → 8 = 23 therefore, n = 3
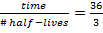 = 12 hours
= 12 hours
- 12.5% =
- Create a half-life chart
| # Half-Lives | % Material | Total Time (hours) |
| 0 | ↓ 100 | 0 |
| 1 | ↓ 50 | ↑ 12 |
| 2 | ↓ 25 | ↑ 24 |
| 3 | 12.5 → | ↑ 36 |